Verzending 24–48 u • Levering in de hele EU • Veilige chemieverpakking
�le und Fette
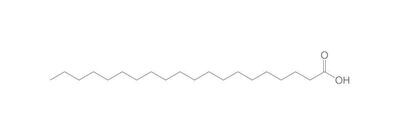
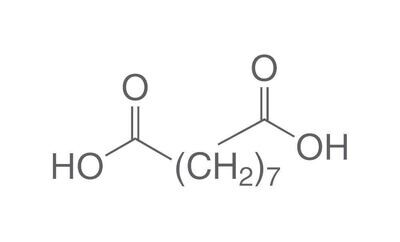
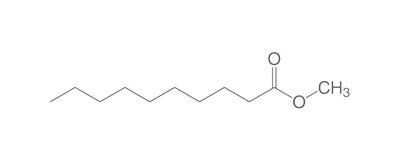
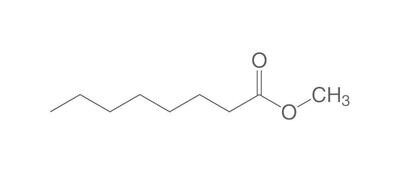
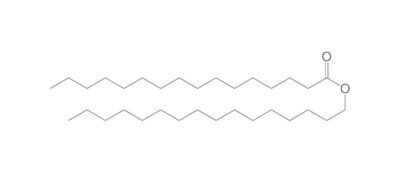
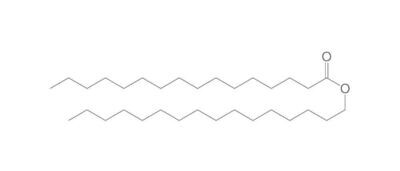
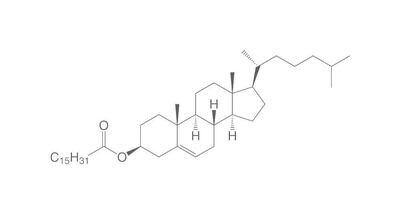
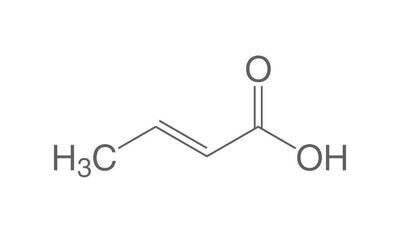
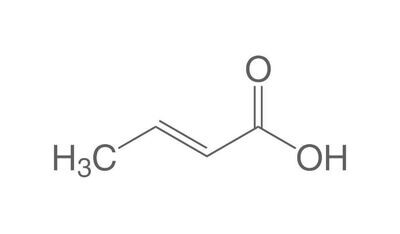
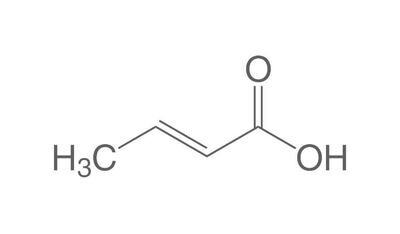
The chemistry of oils and fats is primarily based on their composition, which consists of triglycerides, fatty acids, and glycerol. Let's delve into the chemistry of oils and fats: Triglycerides: Triglycerides are the main constituents of oils and fats. They are esters formed by the condensation of three fatty acid molecules with one glycerol molecule. The esterification process involves the removal of three water molecules, resulting in the formation of the triglyceride molecule. Triglycerides serve as energy storage molecules in plants and animals. Fatty Acids: Fatty acids are the building blocks of triglycerides. They are long hydrocarbon chains with a carboxyl group (COOH) at one end. Fatty acids can be categorized into different types based on their structure: Saturated Fatty Acids: Saturated fatty acids have single bonds between all carbon atoms in their hydrocarbon chain. This results in a straight and rigid structure. Examples include stearic acid and palmitic acid. Unsaturated Fatty Acids: Unsaturated fatty acids contain one or more double bonds in their hydrocarbon chain. These double bonds introduce kinks in the structure, making unsaturated fatty acids more fluid. Monounsaturated fatty acids have one double bond, such as oleic acid, while polyunsaturated fatty acids have multiple double bonds, like linoleic acid and linolenic acid. Trans Fatty Acids: Trans fatty acids are unsaturated fatty acids with a specific configuration of the double bonds. They have a trans configuration, which is artificially produced during the hydrogenation process. Trans fats are known to have adverse health effects. Hydrolysis: Oils and fats can undergo hydrolysis, a chemical reaction in which triglycerides are broken down into glycerol and individual fatty acids. This reaction can be catalyzed by either acids or enzymes known as lipases. Hydrolysis of oils and fats is important in digestion, as lipases in the digestive system break down triglycerides into their components for absorption. Oxidation: Oils and fats are susceptible to oxidation, which can lead to rancidity and degradation of their quality. Oxidation occurs when unsaturated fatty acids react with oxygen in the presence of heat, light, or catalysts. This reaction results in the formation of free radicals and compounds like peroxides, aldehydes, and ketones. Antioxidants are often added to oils and fats to inhibit oxidation and extend their shelf life. Soap Formation: The hydrolysis of oils and fats in the presence of alkalis, such as sodium hydroxide (NaOH) or potassium hydroxide (KOH), leads to the formation of soap. This process is called saponification. The alkali reacts with the fatty acids, releasing glycerol and forming salts of the fatty acids, which are the soap molecules. Understanding the chemistry of oils and fats is crucial in various fields, including food science, nutrition, biochemistry, and industrial applications. By studying their composition and chemical properties, scientists can optimize their use and develop products with desired characteristics and functionality.
Verfeinern nach
Filter
Filter
Anisöl, natürlich, 100 ml, Glasverpackung
Anisöl, natürlich, 100 ml, Glasverpackung
Art.-Nr. 7036.1
€ 43,60

Arachidonsäure, >99 %, 1 g, Glasverpackung
Arachidonsäure, >99 %, 1 g, Glasverpackung
Art.-Nr. 4717.3
€ 758,04

Arachidonsäure, >99 %, 10 mg, Glasverpackung
Arachidonsäure, >99 %, 10 mg, Glasverpackung
Art.-Nr. 4717.2
€ 66,61

Arachidonsäure, >99 %, 100 mg, Glasverpackung
Arachidonsäure, >99 %, 100 mg, Glasverpackung
Art.-Nr. 4717.1
€ 159,91

Avocadoöl, raffiniert, 1 l, Glasverpackung
Avocadoöl, raffiniert, 1 l, Glasverpackung
Art.-Nr. 9341.1
€ 86,19
Baldrianöl, naturrein, 10 ml, Glasverpackung
Baldrianöl, naturrein, 10 ml, Glasverpackung
Art.-Nr. 6588.2
€ 62,01
Baldrianöl, naturrein, 25 ml, Glasverpackung
Baldrianöl, naturrein, 25 ml, Glasverpackung
Art.-Nr. 6588.3
€ 129,41
Baldrianöl, naturrein, 50 ml, Glasverpackung
Baldrianöl, naturrein, 50 ml, Glasverpackung
Art.-Nr. 6588.1
€ 247,33
Cajeputöl, natürlich, 100 ml, Glasverpackung
Cajeputöl, natürlich, 100 ml, Glasverpackung
Art.-Nr. 6105.1
€ 37,40
Cajeputöl, natürlich, 500 ml, Glasverpackung
Cajeputöl, natürlich, 500 ml, Glasverpackung
Art.-Nr. 6105.2
€ 129,41
Campheröl, naturrein, 1 l, Glasverpackung
Campheröl, naturrein, 1 l, Glasverpackung
Art.-Nr. 3357.1
€ 76,50
Endbenutzererklärung
Cassiaöl, chinesisch, natürlich, 100 ml, Glasverpackung
Cassiaöl, chinesisch, natürlich, 100 ml, Glasverpackung
Nur für registrierte Unternehmen
Art.-Nr. 6593.1
€ 56,98
Cedernblätteröl, rein, 100 ml, Glasverpackung
Cedernblätteröl, rein, 100 ml, Glasverpackung
Art.-Nr. 6594.1
€ 116,79
Cubebaöl, natürlich, 100 ml, Glasverpackung
Cubebaöl, natürlich, 100 ml, Glasverpackung
Art.-Nr. 6608.1
€ 37,40

Erdnussöl, raffiniert, 1 l, Glasverpackung
Erdnussöl, raffiniert, 1 l, Glasverpackung
Art.-Nr. 9332.1
€ 35,58

Erdnussöl, raffiniert, 2.5 l, Glasverpackung
Erdnussöl, raffiniert, 2.5 l, Glasverpackung
Art.-Nr. 9332.3
€ 65,49
Estragonöl, rein, 50 ml, Glasverpackung
Estragonöl, rein, 50 ml, Glasverpackung
Art.-Nr. 6602.1
€ 101,81
Fenchelöl, künstlich, 100 ml, Glasverpackung
Fenchelöl, künstlich, 100 ml, Glasverpackung
Art.-Nr. 3299.2
€ 23,60
Fenchelöl, künstlich, 500 ml, Glasverpackung
Fenchelöl, künstlich, 500 ml, Glasverpackung
Art.-Nr. 3299.1
€ 62,01
Preise anzeigen in:EUR